In 1871 he predicted Eka-Aluminum would have an atomic mass of 68 (actual 69.7), a density of 6 gm/cm³ (actual 5.9), a low melting point (actual 29.8☌), and compounds with Oxygen or Chlorine. Mendeleev labeled an unknown element “Eka-Aluminum” because it would fill a gap in the same column as Aluminum but one row down. He could even make predictions about the properties of the undiscovered elements. Thus, he was able to leave gaps for elements, which were unknown at the time. Mendeleev recognized properties that repeated in a predictable, periodic pattern related to an element’s number of electrons. He then proceeded through the known elements sorting by weight and electrons. Hydrogen was the lightest element known, and it had one electron, so it became Element 1 on the Periodic Table. If Mendeleev did not know about protons and neutrons in the atomic nucleus, why does his Periodic Table work? Mendeleev understood the relationship between atomic weights and valence electrons, which gave him an indirect understanding of protons in the nucleus. The valence electrons are held closer towards the nucleus of the atom.Learn more about elements: The Secret Lives of the Elements Why does Mendeleev’s Periodic Table Work? 
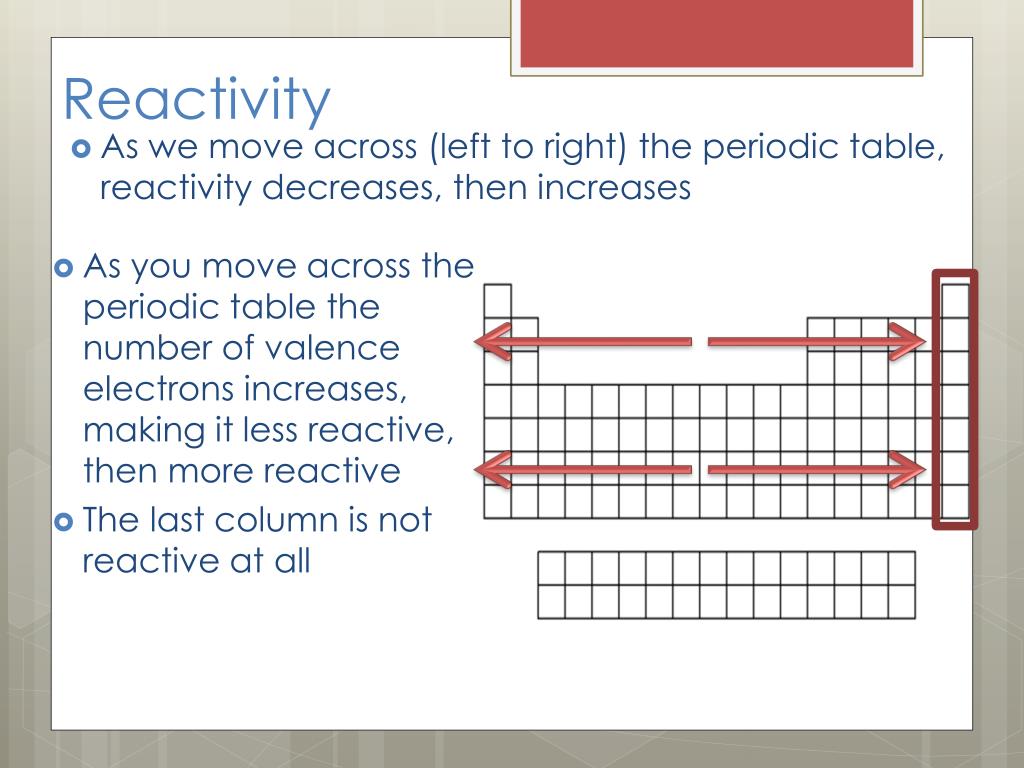
This means that the nucleus attracts the electrons more strongly, pulling the atom's shell closer to the nucleus. The effect of increasing proton number is greater than that of the increasing electron number therefore, there is a greater nuclear attraction. However, at the same time, protons are being added to the nucleus, making it more positively charged. This is because, within a period or family of elements, all electrons are added to the same shell. Atomic radius patterns are observed throughout the periodic table.Ītomic size gradually decreases from left to right across a period of elements. 
The covalent radii of these molecules are often referred to as atomic radii. Nevertheless, it is possible for a vast majority of elements to form covalent molecules in which two like atoms are held together by a single covalent bond. Some are bound by covalent bonds in molecules, some are attracted to each other in ionic crystals, and others are held in metallic crystals. However, this idea is complicated by the fact that not all atoms are normally bound together in the same way. The atomic radius is one-half the distance between the nuclei of two atoms (just like a radius is half the diameter of a circle). This is caused by the increase in atomic radius.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
